Pharmaceutical manufacturers and dispensers participated in a digital identity pilot that created secure ATP credentials and opened new, direct communication channels between disparate trading partners.

Read the full XATP Working Group whitepaper at xatp.org. The XATP app is available for dispensers now in the Apple App Store.
CHALLENGE
Pharmaceutical supply chain stakeholders face new and emerging requirements under the Drug Supply Chain Security Act (DSCSA). One such requirement is an example of a “know your customer” requirement, in which each Authorized Trading Partner (ATP) is required to confirm that their trading partners are also authorized. This places a significant burden on the first and last miles of the supply chain, as tens of thousands of dispensers send verification requests to manufacturers and repackagers who are required to respond within 24 hours, even when there is no direct business relationship. This must be achieved without breaching the strict personal information mandates of privacy laws such as CalPrivacy and GDPR.
OPPORTUNITY
The near-universal adoption of GS1 barcodes and unique identifiers on drug packages in the United States has created a common data model for the entire US pharmaceutical supply chain, with opportunities for manufacturers and dispensers to unlock shared business value and drive better patient outcomes.
Dispensers:
- barcodes can be scanned by off-the-shelf iPhones (no custom hardware required)
- fast and secure verification for any drug in the US
- up-to-date master data, including recalls, updated expiration dates, and med guides
- product verification certificates support saleable returns and custody transfer
Manufacturers:
- unprecedented visibility into the last mile of product distribution
- enhanced counterfeit detection with statistically significant sample data
- enhanced shortage mitigation with real-time market intelligence
- enhanced relationships with indirect trading partners
SOLUTION
XATP is a framework for ATP authentication, verification routing, and saleable returns documentation. In a pilot launched by a working group with manufacturers and dispensers, XATP proved capable of enabling secure communication among ATPs by onboarding ATPs and their representatives, and issuing them verifiable ATP credentials following the validation of their licenses.
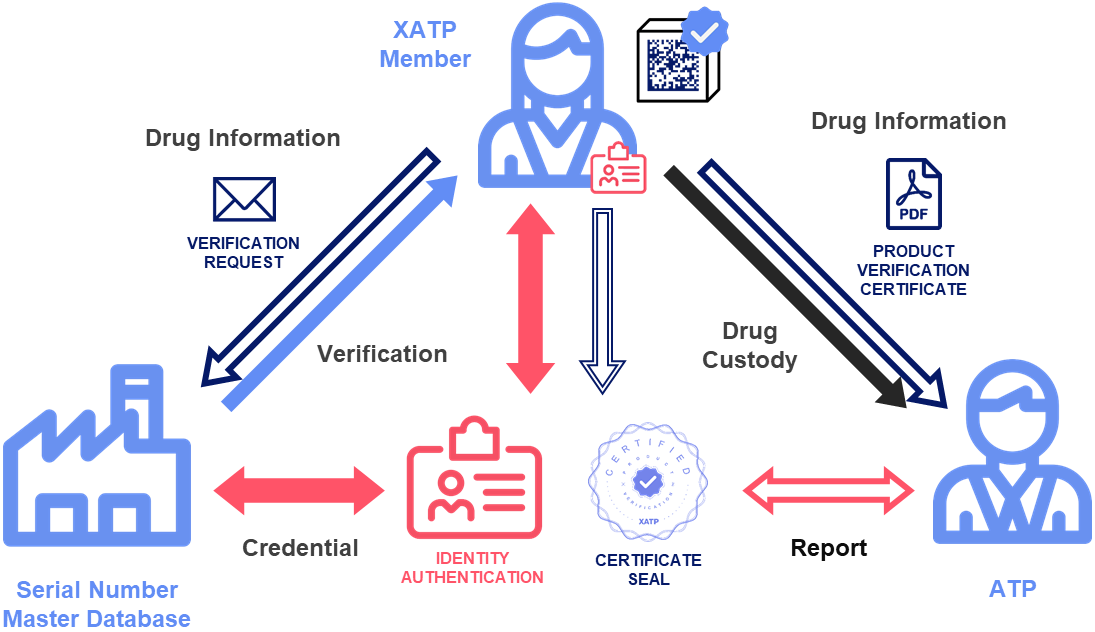
A dispenser with an iPhone and an acceptable form of ID can be remotely authenticated as an ATP, scan the barcode from a drug in their hand, and use the XATP app to send a verification request. This request, which pulls the GTIN from the scan, is used to identify the appropriate point of contact (POC) for the manufacturer or repackager, and sends them a secure email. Pilot participants successfully verified test barcodes by clicking one of two buttons in the email (verified or not verified).
Only dispensers require XATP accounts, as other ATPs can authenticate identities and verify drugs using widely adopted technologies.
This response contains a certificate that carries the digital POC ATP credential, and can be used to generate supporting documentation — a certificate of authenticity — for transactions involving third-party ATPs such as saleable returns. Only dispensers require XATP accounts, as other ATPs can authenticate identities and verify drugs using widely adopted technologies.
NEXT STEPS
The XATP framework mitigates risk for stakeholders to rapidly attain compliance with DSCSA obligations such as enhanced drug verification, and sets a path to compliance with the full set of traceability requirements in 2023. XATP is now available for dispensers to try in the Apple App Store, with scaled deployments in Q1 2021.
To unlock real-time verification, robust business intelligence, and greater security, manufacturers only need to integrate an XATP machine-to-machine (M2M) gateway with their serialization database. The XATP M2M gateway hosts the manufacturer’s identity credential and automatically processes verification requests from the XATP network and other VRS providers, making interoperability easy.
XATP WORKING GROUP
Our fellow pilot members included Natalie Helms, Alina Grigorian & Nikkhil Vinnakota (Amgen); Nirmal Annamreddy, Kathy Daniusis, Mark Karhoff & Vid Rajaram (Genentech); Greg Plante (IQVIA); Todd Barrett (Providence Health); Arthi Nagaraj (Sanofi); Jess de Jesus, Ghada Ashkar & Kalpan Patel (UCLA Health); and LedgerDomain’s own Will Chien & Will Jack.
Special thanks also to Jose Arrieta (formerly US Department of Health and Human Services); Paul Hackett (Accenture); Diane Shoda (Greyscaling); Alan Lodder (formerly Intermountain); Mike Karhoff (Ten Count Consulting); Mike Marchant (UC Davis); Jen Colon (Yale); and LedgerDomain’s own Dr. Leo Alekseyev, Rick Burgess, Alex Colgan, Dr. Victor Dods, Mike Lodder & Ben Nichols.
Want to learn more? Read the full whitepaper at xatp.org and contact admin@xatp.org.

